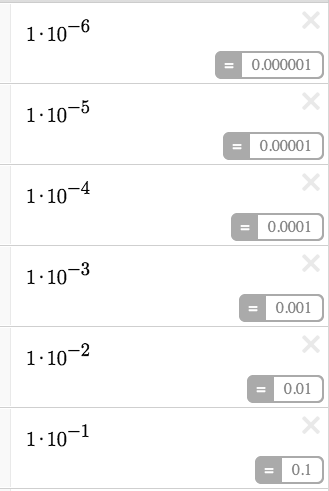
The equations below reflect BOTH [OH-]=1x10^(-pOH), on the pOH scale for bases and [H+]=1x10^(-pH) on the pH scale for acids
Homework
Spelling 8: test tomorrow
BWHTW: chapters 7 & 8 quiz
SS: Europe, Africa, & Asia maps (label)
Algebra 1: p. 71, #45-56 & test redos
Summer vocab #1 quiz tomorrow
Math 7: worksheets (3 graphing worksheets)
Spelling 8: test tomorrow
BWHTW: chapters 7 & 8 quiz
SS: Europe, Africa, & Asia maps (label)
Algebra 1: p. 71, #45-56 & test redos
Summer vocab #1 quiz tomorrow
Math 7: worksheets (3 graphing worksheets)

 RSS Feed
RSS Feed